PRODUCT CENTER
特色產(chǎn)品當前位置:首頁 > 特色產(chǎn)品
肥胖癥是美國第二大可預防死亡原因。它與多種合并癥相關,包括2型糖尿病(T2D)、代謝功能障礙相關肝病(MASLD,之前稱為非酒精性脂肪肝病,NAFLD)、心血管疾病和癌癥。最初為治療T2D而開發(fā)的胰高血糖素樣肽1(GLP-1)、葡萄糖依賴性胰島素促進多肽(GIP)和胰高血糖素受體激動劑在肥胖癥治療中發(fā)現(xiàn)了新的治療用途。
Cayman中國區(qū)總代理,艾美捷科技,提供這些僅供研究使用的試劑,以支持2型糖尿病和肥胖癥等代謝性疾病的研究。本文探討了這些試劑背后的科學。
胰島素和胰高血糖素調(diào)節(jié)血糖穩(wěn)態(tài)
調(diào)節(jié)血糖水平主要由胰腺釋放的激素:胰島素和胰高血糖素驅(qū)動。這兩種激素作用相反,共同調(diào)節(jié)血糖穩(wěn)態(tài)。
胰島素在血糖水平高(高血糖)時釋放,通過向細胞發(fā)出信號吸收血液中的葡萄糖以供能量或儲存,從而降低血糖水平。胰高血糖素在血糖水平低(低血糖)時釋放。它刺激肝臟通過糖原分解增加葡萄糖產(chǎn)生,從而提高血糖水平。
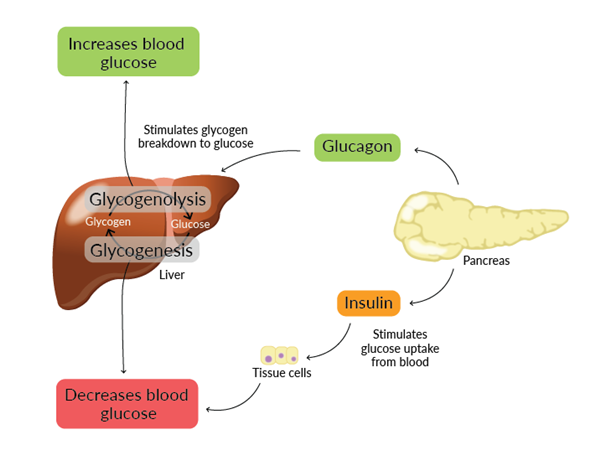
圖1. 葡萄糖穩(wěn)態(tài)中胰島素和胰高血糖素途徑的總結。
GLP-1和GIP通過刺激餐后胰島素釋放來調(diào)節(jié)血糖水平
餐后血糖水平通常會上升,禁食時下降。餐后,葡萄糖——細胞進行無數(shù)生物過程所需的關鍵能量來源——被釋放到血液中,增加血糖水平。
為了刺激餐后胰島素的分泌,身體利用兩種在腸道產(chǎn)生的腸促胰島素激素:胰高血糖素樣肽1(GLP-1)和葡萄糖依賴性胰島素促進多肽(GIP)。這些激素在餐后釋放,并刺激胰腺分泌胰島素,向細胞發(fā)出信號吸收葡萄糖以供能量或儲存,減少餐后血糖水平的增加。
GLP-1調(diào)節(jié)食欲和食物攝入量
除了刺激胰島素產(chǎn)生外,GLP-1(而不是GIP)還減緩胃排空,降低腸道運動性,減少食物攝入和食欲,并通過復雜的中樞和外周途徑刺激胰高血糖素依賴的能量消耗。這些效應共同促進減重,使GLP-1受體(GLP-1R)激動劑成為肥胖癥的有價值方法。
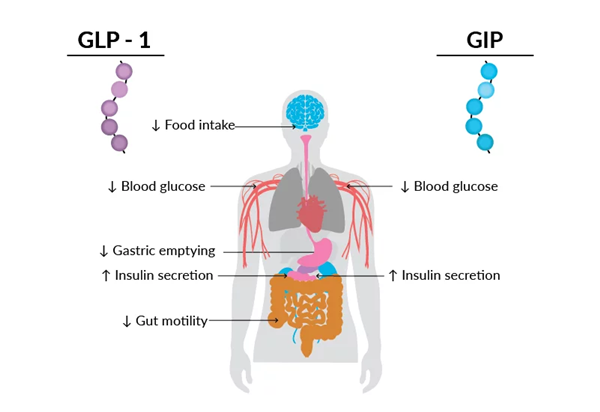
圖2. GLP-1和GIP在葡萄糖穩(wěn)態(tài)和體重減輕中的作用的總結
肥胖癥研究中的腸促胰島素基礎療法
單一激動劑
塞美格魯肽是一種GLP-1R激動劑,是一種GLP-1的肽類似物,用于促進減重。研究表明,與僅改變生活方式(如減少熱量攝入和增加體力活動)相比,它可以幫助肥胖個體實現(xiàn)更大的減重效果。
其他基于肽的GLP-1R激動劑,如利西那肽、利拉魯肽、度拉糖肽和艾塞那肽,它們在分子結構、大小、藥理學、效力和安全性上有所不同,根據(jù)個體情況提供各種優(yōu)勢。
非肽類療法可能比基于肽的療法提供優(yōu)勢。基于肽的療法通常是大分子量化合物,需要注射,而非肽類GLP-1R激動劑,如orforglipron,或正向別構調(diào)節(jié)劑,如V-0219,可以作為口服制劑。這些非肽GLP-1R激動劑正在進行臨床試驗。
雙重激動劑
曲普瑞肽是一種首創(chuàng)的雙重GLP-1R和GIPR激動劑。雖然GIP單獨似乎不會延遲胃排空或改變食物攝入和食欲,但由于尚不清楚的原因,GLP-1和GIP受體(GIPR)激動劑協(xié)同作用,與單獨使用GLP-1R激動劑相比提供更大的益處。這些雙重GLP-1R和GIPR激動劑,俗稱“雙促胰島素”,似乎也因為它們的雙重作用機制而減少副作用。這些雙重激動劑的優(yōu)勢激發(fā)了進一步研究針對多重作用機制的療法。
三重激動劑
一種新的方法是通過針對三個激素受體。胰高血糖素受體(GCGR)拮抗劑最初作為T2D療法被追求,基于它們會減弱葡萄糖產(chǎn)生并促進胰島素分泌的想法。然而,由于與肝脂肪變性發(fā)展相關的MASLD安全問題,這些藥物開發(fā)努力不再受青睞。
然而,盡管GCGR激動劑具有高血糖效應,但它們通過刺激熱生成促進飽腹感和增加能量消耗,使它們成為研究肥胖癥的潛在寶貴補充。GCGR激動作用的高血糖效應可以通過包括刺激胰島素分泌的制劑來抵消,從而降低血糖水平,使GLP-1R和/或GIPR激動劑的多激動劑制劑成為肥胖癥的新療法。事實上,retatrutide,一種三重GLP-1R、GIPR和GCGR激動劑,在臨床試驗中顯示出前景。
| GLP-1R Agonists | GIPR Agonists | GCGR Agonists | |
| SemaglutideA peptide analog |
| ||
| LixisenatideA peptide analog |
| ||
| DulaglutideA peptide analog |
| ||
| ExendinA peptide analog |
| ||
| OrforglipronA non-peptide agonist |
| ||
| V-0219A non-peptide positive allosteric modulator |
| ||
| LSN3318839A non-peptide positive allosteric modulator |
| ||
| Taspoglutide (acetate)A peptide analog |
| ||
| LiraglutideA peptide analog |
| ||
| TirzepatideA peptide analog |
|
| |
| BamadutideA peptide analog |
|
| |
| Cotadutide (acetate)A peptide analog |
|
| |
| SurvodutideA peptide analog |
|
| |
| MazdutideA peptide analog |
|
| |
| RetatrutideA peptide analog |
|
|
|
| SAR441255 (sodium salt)A peptide analog |
|
|
|
| These products are for scientific research use only. | |||
未來方向
此外,這些重新用于T2D治療的許多療法不僅在肥胖癥中被探索,還在與肥胖癥相關的其他疾病中被探索,包括心力衰竭和心血管疾病。這些相同的制劑還顯示出治療其他疾病,如物質(zhì)使用障礙和神經(jīng)退行性疾病如阿爾茨海默病和帕金森病的潛在益處。
以上產(chǎn)品僅供科學研究使用。
超900+糖尿病研究相關研究試劑,歡迎垂詢Cayman中國區(qū)總代理,艾美捷科技:
參考文獻
1. Wang, Y., Beydoun, M.A., Min, J., et al. Has the prevalence of overweight, obesity and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int. J. Epidemiol.49(3), 810-823 (2020).
2. Zhang, X., Ha, S., Lau, H. C.-H., et al. Excess body weight: Novel insights into its roles in obesity comorbidities. Semin. Cancer Biol.92, 16-27 (2023).
3. R?der, P.V., Wu, B., Liu, Y., et al. Pancreatic regulation of glucose homeostasis. Exp. Mol. Med.48(3), e219 (2016).
4. Nakrani, M.N., Wineland, R.H., and Anjum, F. Physiology, glucose metabolism. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing (2024). Available from: https://www.ncbi.nlm.nih.gov/books/NBK560599/
5. Baggio, L.L. and Drucker, D.J. Biology of incretins: GLP-1 and GIP. Gastroenterology132(6), 2131-2157 (2007).
6. Nauck, M.A., Quast, D.R., Wefers, J. et al. The evolving story of incretins (GIP and GLP-1) in metabolic and cardiovascular disease: A pathophysiological update. Diabetes Obes. Metab.23(Suppl 3), 5-29 (2021).
7. González-García, I., Milbank, E., Diéguez, C., et al. Glucagon, GLP-1 and thermogenesis. Int. J. Mol. Sci.20(14), 3445 (2019).
8. Holst, J.J. and Rosenkilde, M.M. GIP as a therapeutic target in diabetes and obesity: Insight from incretin co-agonists. J. Clin. Endocrinol. Metab.105(8), e2710-e2716 (2020).
9. Edholm, T., Degerblad, M., Gryb?ck, P. et al. Differential incretin effects of GIP and GLP-1 on gastric emptying, appetite, and insulin-glucose homeostasis. Neurogastroenterol. Motil.22(11), 1191-1200, e315 (2010).
10. Wadden, T.A., Chao, A.M., Moore, M., et al. The role of lifestyle modification with second-generation anti-obesity medications: Comparisons, questions, and clinical opportunities. Curr. Obes. Rep.12(4), 453-473 (2023).
11. Almandoz, J.P., Lingvay, I., Morales, J., et al. Switching between glucagon-like peptide-1 receptor agonists: Rationale and practical guidance. Clin. Diabetes38(4), 390-402 (2020).
12. Malik, F. and Li, Z. Non‐peptide agonists and positive allosteric modulators of glucagon‐like peptide‐1 receptors: Alternative approaches for treatment of type 2 diabetes. Br. J. Pharmacol.179(4), 511-525 (2022).
13. Wharton, S., Blevins, T., Connery, L., et al. Daily oral GLP-1 receptor agonist orforglipron for adults with obesity. N. Engl. J. Med.389(10), 877-888 (2023).
14. Decara, J.M., Vázquez-Villa, H., Brea, J., et al. Discovery of V-0219: A small-molecule positive allosteric modulator of the glucagon-like peptide-1 receptor toward oral treatment for "diabesity"'. J. Med. Chem.65(7), 5449-5461 (2022).
15. Lin, F., Yu, B., Ling, B., et al. Weight loss efficiency and safety of tirzepatide: A systematic review. PLoS One18(5), e0285197 (2023).
16. Hope, D.C.D., Vincent, M.L., and Tan, T.M.M. Striking the balance: GLP-1/glucagon co-agonism as a treatment strategy for obesity. Front. Endocrinol.(Lausanne)12, 735019 (2021).
17. Novikoff, A. and Müller, T.D. The molecular pharmacology of glucagon agonists in diabetes and obesity. Peptides165, 171003 (2023).
18. Naeem, M., Imran, L., and Banatwala, U.E.S.S. Unleashing the power of retatrutide: A possible triumph over obesity and overweight: A correspondence. Health Sci. Rep.7(2), e1864 (2024).
19. Kosiborod, M.N, Abildstr?m, S.Z., Borlaug, B.A., et al. Semaglutide in patients with heart failure with preserved ejection fraction and obesity. N. Engl. J. Med.389(12), 1069-1084 (2023).
20. Husain, M., Bain, S.C., Jeppesen, O.K., et al. Semaglutide (SUSTAIN and PIONEER) reduces cardiovascular events in type 2 diabetes across varying cardiovascular risk. Diabetes Obes. Metab.22(3), 442-451 (2020).
21. Ryan, D.H., Lingvay, I., Colhoun, H.M., et al. Semaglutide effects on cardiovascular outcomes in people with overweight or obesity (SELECT) rationale and design. Am. Heart J.229, 61-69 (2020).
22. Edwards, K.L. and Minze, M.G. Dulaglutide: An evidence-based review of its potential in the treatment of type 2 diabetes. Core Evid.10, 11-21 (2015).
23. Thomsen, M., Holst, J.J., Molander, A., et al. Effects of glucagon-like peptide 1 analogs on alcohol intake in alcohol-preferring vervet monkeys. Psychopharmacology(Berl.)236(2), 603-611 (2019).
24. Yammine, L., Green, C.E., Kosten, T.R., et al. Exenatide adjunct to nicotine patch facilitates smoking cessation and may reduce post-cessation weight gain: A pilot randomized controlled trial. Nicotine Tob. Res.23(10), 1682-1690 (2021).
25. Leggio, L., Hendershot, C.S., Farokhnia, M., et al. GLP-1 receptor agonists are promising but unproven treatments for alcohol and substance use disorders. Nat. Med.29(12), 2993-2995 (2023).
26. Mahapatra, M.K., Karuppasamy, M., and Sahoo, B.M. Therapeutic potential of semaglutide, a newer GLP-1 receptor agonist, in abating obesity, non-alcoholic steatohepatitis and neurodegenerative diseases: A narrative review. Pharm. Res.39(6), 1233-1248 (2022).
歡迎垂詢,Cayman 中國區(qū)總代理,艾美捷科技,m.zihai029.com,了解更多產(chǎn)品詳情,開啟糖尿病與肥胖研究的新篇章。


微信掃碼在線客服