AGENT BRAND
品牌專題IFN-α(Interferon-alpha,IFN-Alpha)是指干擾素α,它是一種類型的細(xì)胞因子,屬于干擾素家族。干擾素是哺乳動(dòng)物細(xì)胞因子家族,具有調(diào)節(jié)免疫反應(yīng)和抗病毒能力的作用。它們由大多數(shù)細(xì)胞類型合成和分泌,以響應(yīng)病原體。除了抗病毒特性外,干擾素還被證明具有抗增殖、免疫調(diào)節(jié)和許多其他活性。
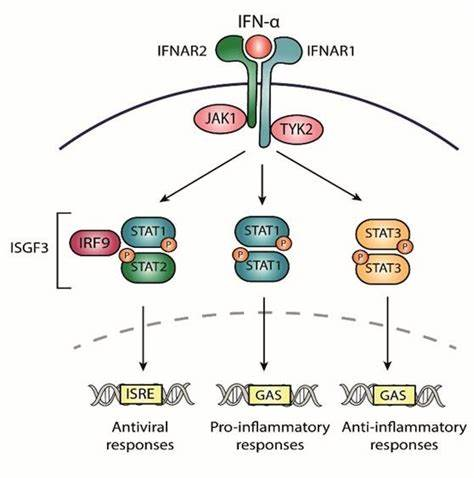
在人類中,IFN-α由一組氨基酸序列同源性大于85%的蛋白質(zhì)組成。已鑒定出許多個(gè)人類IFN-α亞型;許多顯示不同的屬性。目前尚不清楚為什么有多種IFN-α亞型。各種研究表明,它們具有重疊但又獨(dú)特的生物活性。 我們推薦高靈敏度的人IFN-α全亞型ELISA試劑盒旨在對(duì)自身免疫血清、正常血清、EDTA 血漿和組織培養(yǎng)基(TCM)中的所有人IFN-α亞型進(jìn)行準(zhǔn)確的低 pg/ml 測(cè)量。提供了一種可靠且靈敏的方法,因此對(duì)于免疫學(xué)、炎癥和治療藥物監(jiān)測(cè)方面的研究非常有價(jià)值。作為PBL Assay Science在中國(guó)區(qū)域的代理商,艾美捷科技將為中國(guó)客戶提供全面的PBL Assay Science產(chǎn)品以及客戶訂制化服務(wù)。歡迎大家隨時(shí)聯(lián)系我們。

| 產(chǎn)品貨號(hào) | 產(chǎn)品名稱 | 樣本類型 |
| 41115 | Human IFN-Alpha All Subtype ELISA Kit, High Sensitivity (Serum, Plasma, TCM) | 血清,血漿,組織培養(yǎng)基 |
| 高靈敏度的人IFN-α全亞型ELISA試劑盒(貨號(hào):41115) | |
| 樣品類型 | 血清、血漿、組織培養(yǎng)基 |
| 檢測(cè)范圍 | 1.95 - 125 pg/ml |
| 靈敏度(LLOQ) | 1.95 pg/ml |
| 特異性 | 人IFN-α,檢測(cè)所有12種IFN-α亞型,以準(zhǔn)確地測(cè)量樣品的總量。 |
| 變異系數(shù)和加標(biāo)回收率 | Inter-Assay: ≤ 10% Intra-Assay: ≤ 8% Spike Recovery: ≥ 80% in Serum |
| 該試劑盒測(cè)試的正常人血清中干擾素α各亞型的代表性定量下限和檢測(cè)下限(pg/ml) | 專為檢測(cè)人類所有IFN-α亞型而設(shè)計(jì)。
|
| Protocol |
|
| 典型標(biāo)準(zhǔn)曲線 |
|
| 正常人血清中IFN-α亞型標(biāo)準(zhǔn)曲線 |
|
引用文獻(xiàn)(Citations):
Ou, B.S. et al., (2024), "Nanoparticle-Conjugated Toll-Like Receptor 9 Agonists Improve the Potency, Durability, and Breadth of COVID-19 Vaccines", ACS NanoDOI: 10.1021/acsnano.3c09700
Biava, M. et al., (2023), "In Vitro and In Vivo Crosstalk between Type I IFN and IL-8 Responses in SARS-CoV-2 Infection", Microorganisms, 11(11):2787, PMID: 38004798, DOI: 10.3390/microorganisms11112787
Grunhagel, B., et al., (2023), "Reduction of IFN-I Responses by Plasmacytoid Dendritic Cells in a Longitudinal Trans Men Cohort, iScience, DOI: 10.1016/j.isci.2023.108209
Nagaoka, K. et al., (2023), "Dominant CT Patterns and Immune Responses during the Early Infection Phases of Different SARS-CoV-2 Variants", Viruses, 15:1304, DOI: 10.3390/v15061304
Kida, Y. et al., (2023), "Lethal Interstitial Lung Disease Associated with a Gain-of-Function Mutation in IFIH1", Clin Immunol., PMID: 37126154, DOI: 10.1007/s10875-023-01494-8
Kim, S.T. et al., (2023). "Interferon and interferon-induced cytokines as markers of impending clinical progression in ANA+ individuals without a systemic autoimmune rheumatic disease diagnosis", Arthritis Res. Ther., PMID: 36765391, DOI: 10.1186/s13075-023-02997-w
Bibby, J.A., et al., (2022), "Systematic single-cell pathway analysis to characterize early T cell activation, Cell Rep., 41(8):111697, PMID: 36417885, DOI: 10.1016/j.cellrep.2022.111697
Rajamanickam, A. et al., (2022), "Restoration of dendritic cell homeostasis and Type I/Type III interferon levels in convalescent COVID-19 individuals, BMC Immunol., 23(1):51, PMID: 36289478, DOI: 10.1186/s12865-022-00526-z
Kubo, S. et al., (2022), "Lactoferrin and its digestive peptides induce interferon-a production and activate plasmacytoid dendritic cells ex vivo", Biometals, PMID: 36018422, DOI: 10.1007/s10534-022-00436-y
Nagaoka, N., et al., (2022), "Effect of Casirivimab/Imdevimab Treatment on Serum Type I Interferon Levels in SARS-CoV-2 Infection", Viruses, 14(7):1399, DOI: 10.3390/v14071399
Edahiro, Y., Ohishi, K., Gotoh, A. et al., (2022), "Efficacy and safety of ropeginterferon alfa-2b in Japanese patients with polycythemia vera: an open-label, single-arm, phase 2 study", J. Hematol., PMID: 35430707, DOI: 10.1007/s12185-022-03341-9
Kanazawa, N. et al.,(2021), Heterozygous missense variant of the proteasome subunit β-type 9 causes neonatal-onset autoinflammation and immunodeficiency, Nature Communications, 12:6819, DOI: 10.1038/s41467-021-27085-y
Jablonska, A., et al., (2021), The TLR92848C/T Polymorphism Is Associated with the CMV DNAemia among HIV/CMV Co-Infected Patients, Cells, 10:2360, DOI: 10.3390/cells10092360,
Peluso, Michael, et al.(2020). Liver function test abnormalities in a longitudinal cohort of Thai individuals treated since acute HIV infection. PLOS Pathogens, 9 pgs. PMID: 31953919.
Dagenais-Lussier, Xavier, et al.(2019). USP18 is a significant driver of memory CD4 T-cell reduced viability caused by type I IFN signaling during primary HIV-1 infection. PLOS Pathogens, 32 pgs. PMID: 31658294.
Colavita, Francesca, et al.(2018). Overproduction of IL-6 and Type-I IFN in a Lethal Case of Chikungunya Virus Infection in an Elderly Man During the 2017 Italian Outbreak. Open Forum Infectious Diseases, 21 pgs. PMID: 30539034.
Williams, Dionna, et al.(2018). CCR2 Signaling Selectively Regulates IFN-alpha: Role of Beta-Arrestin 2 in IFNAR1 Internalization. Journal of Immunology, 19 pgs. PMID: 30504423.
Zhang, Guoliang, et al.(2018). A Proline Deletion in IFNAR1 Impairs IFN-Signaling and Underlies Increased Resistance to Tuberculosis in Humans. Nature Communications, 9 pgs. PMID: 29311663.
Murayama, Goh, et al. (2017). Enhanced IFN-alpha Production is Associated with Increased TLR7 Retention in the Lysosomes of Palasmacytoid Dendritic Cells in Systemic Lupus Erythematosus. Arthritis Research & Therapy, 11 pgs. PMID: 29052537.
Wither, Joan, et al. (2017). Presence of an Interferon Signature in Individuals Who are Anti-Nuclear Antibody Positive Lacking a Systemic Autoimmune Rheumatic Disease Diagnosis. Arthritis Research & Therapy, 11 pgs. PMID: 28245862.
Vanheule, Vincent, et al. (2016). Basic Chemokine-Derived Glycosaminoglycan Binding Peptides Exert Antiviral Properties Against Dengue Virus Serotype 2, Herpes Simplex Virus-1, and Respiratory Syncytial Virus. Biochemical Pharmacology, 13 pgs. PMID: 26551597.
Combes, Alexis, et al. (2020). Global Absence and Targeting of Protective Immune States in Severe COVID-19. Nature, 36 pgs.
看到這兒,您心動(dòng)了嗎?馬上聯(lián)系小艾吧!
美國(guó)的PBL Assay Science(又名:Pestka Biomedical Laboratories, Inc.)成立于1990年,創(chuàng)始人Sidney Pestka被稱為“干擾素之父”,PBL Assay Science 作為干擾素和細(xì)胞因子蛋白和抗體以及預(yù)包裝的一流的干擾素和細(xì)胞因子免疫測(cè)定試劑盒的高質(zhì)量制造商而享有盛譽(yù)。生產(chǎn)和銷售的高品質(zhì)干擾素產(chǎn)品和生物標(biāo)志物檢測(cè)試劑盒在很多高影響力出版物中都有引用,并已在具有挑戰(zhàn)性的樣本中進(jìn)行了外部驗(yàn)證。
PBL Assay Science提供各種干擾素亞型的蛋白,抗體和檢測(cè)試劑盒,如:IFN-Alpha(IFN-ɑ 2a,IFN-ɑ 2b,IFN-ɑ 5,IFN-ɑ 6,IFN-ɑ 7,IFN-ɑ 14,IFN-ɑ 16,IFN-ɑ 17......),IFN-Beta(IFN-β 1a,IFN-β 1b),IFN-Lambda(IFN-λ),IFN-Omega(IFN-ω),IFN-Gamma(IFN-γ)......
武漢艾美捷科技有限公司,簡(jiǎn)稱艾美捷,擁有進(jìn)出口資質(zhì)和自己的國(guó)際物流,與國(guó)內(nèi)外90余家知名的試劑、原料及技術(shù)服務(wù)供應(yīng)商深度合作,是國(guó)內(nèi)生物制藥企業(yè)、診斷企業(yè)、跨國(guó)藥企、各大高校院所采購(gòu)平臺(tái)的首選定供應(yīng)商。

微信掃碼在線客服