PRODUCT CENTER
特色產品IFN-α(Interferon-alpha,IFN-Alpha)是指干擾素α,它是一種類型的細胞因子,屬于干擾素家族。干擾素是哺乳動物細胞因子家族,具有調節(jié)免疫反應和抗病毒能力的作用。它們由大多數細胞類型合成和分泌,以響應病原體。除了抗病毒特性外,干擾素還被證明具有抗增殖、免疫調節(jié)和許多其他活性。
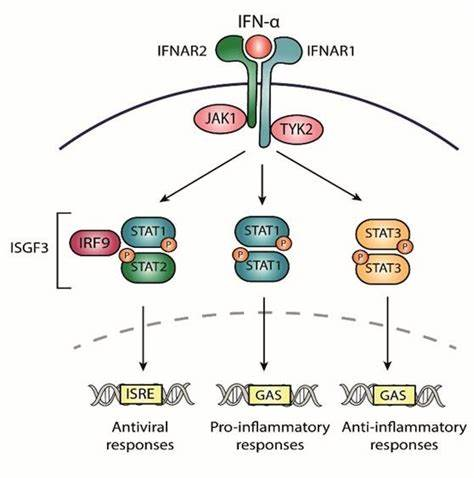
在人類中,IFN-α由一組氨基酸序列同源性大于85%的蛋白質組成。已鑒定出許多個人類IFN-α亞型;許多顯示不同的屬性。目前尚不清楚為什么有多種IFN-α亞型。各種研究表明,它們具有重疊但又獨特的生物活性。 我們推薦高靈敏度的人IFN-α全亞型ELISA試劑盒旨在對自身免疫血清、正常血清、EDTA 血漿和組織培養(yǎng)基(TCM)中的所有人IFN-α亞型進行準確的低 pg/ml 測量。提供了一種可靠且靈敏的方法,因此對于免疫學、炎癥和治療藥物監(jiān)測方面的研究非常有價值。作為PBL Assay Science在中國區(qū)域的代理商,艾美捷科技將為中國客戶提供全面的PBL Assay Science產品以及客戶訂制化服務。歡迎大家隨時聯系我們。

| 產品貨號 | 產品名稱 | 樣本類型 |
| 41115 | Human IFN-Alpha All Subtype ELISA Kit, High Sensitivity (Serum, Plasma, TCM) | 血清,血漿,組織培養(yǎng)基 |
| 高靈敏度的人IFN-α全亞型ELISA試劑盒(貨號:41115) | |
| 樣品類型 | 血清、血漿、組織培養(yǎng)基 |
| 檢測范圍 | 1.95 - 125 pg/ml |
| 靈敏度(LLOQ) | 1.95 pg/ml |
| 特異性 | 人IFN-α,檢測所有12種IFN-α亞型,以準確地測量樣品的總量。 |
| 變異系數和加標回收率 | Inter-Assay: ≤ 10% Intra-Assay: ≤ 8% Spike Recovery: ≥ 80% in Serum |
| 該試劑盒測試的正常人血清中干擾素α各亞型的代表性定量下限和檢測下限(pg/ml) | 專為檢測人類所有IFN-α亞型而設計。
|
| Protocol |
|
| 典型標準曲線 |
|
| 正常人血清中IFN-α亞型標準曲線 |
|
引用文獻(Citations):
Ou, B.S. et al., (2024), "Nanoparticle-Conjugated Toll-Like Receptor 9 Agonists Improve the Potency, Durability, and Breadth of COVID-19 Vaccines", ACS NanoDOI: 10.1021/acsnano.3c09700
Biava, M. et al., (2023), "In Vitro and In Vivo Crosstalk between Type I IFN and IL-8 Responses in SARS-CoV-2 Infection", Microorganisms, 11(11):2787, PMID: 38004798, DOI: 10.3390/microorganisms11112787
Grunhagel, B., et al., (2023), "Reduction of IFN-I Responses by Plasmacytoid Dendritic Cells in a Longitudinal Trans Men Cohort, iScience, DOI: 10.1016/j.isci.2023.108209
Nagaoka, K. et al., (2023), "Dominant CT Patterns and Immune Responses during the Early Infection Phases of Different SARS-CoV-2 Variants", Viruses, 15:1304, DOI: 10.3390/v15061304
Kida, Y. et al., (2023), "Lethal Interstitial Lung Disease Associated with a Gain-of-Function Mutation in IFIH1", Clin Immunol., PMID: 37126154, DOI: 10.1007/s10875-023-01494-8
Kim, S.T. et al., (2023). "Interferon and interferon-induced cytokines as markers of impending clinical progression in ANA+ individuals without a systemic autoimmune rheumatic disease diagnosis", Arthritis Res. Ther., PMID: 36765391, DOI: 10.1186/s13075-023-02997-w
Bibby, J.A., et al., (2022), "Systematic single-cell pathway analysis to characterize early T cell activation, Cell Rep., 41(8):111697, PMID: 36417885, DOI: 10.1016/j.cellrep.2022.111697
Rajamanickam, A. et al., (2022), "Restoration of dendritic cell homeostasis and Type I/Type III interferon levels in convalescent COVID-19 individuals, BMC Immunol., 23(1):51, PMID: 36289478, DOI: 10.1186/s12865-022-00526-z
Kubo, S. et al., (2022), "Lactoferrin and its digestive peptides induce interferon-a production and activate plasmacytoid dendritic cells ex vivo", Biometals, PMID: 36018422, DOI: 10.1007/s10534-022-00436-y
Nagaoka, N., et al., (2022), "Effect of Casirivimab/Imdevimab Treatment on Serum Type I Interferon Levels in SARS-CoV-2 Infection", Viruses, 14(7):1399, DOI: 10.3390/v14071399
Edahiro, Y., Ohishi, K., Gotoh, A. et al., (2022), "Efficacy and safety of ropeginterferon alfa-2b in Japanese patients with polycythemia vera: an open-label, single-arm, phase 2 study", J. Hematol., PMID: 35430707, DOI: 10.1007/s12185-022-03341-9
Kanazawa, N. et al.,(2021), Heterozygous missense variant of the proteasome subunit β-type 9 causes neonatal-onset autoinflammation and immunodeficiency, Nature Communications, 12:6819, DOI: 10.1038/s41467-021-27085-y
Jablonska, A., et al., (2021), The TLR92848C/T Polymorphism Is Associated with the CMV DNAemia among HIV/CMV Co-Infected Patients, Cells, 10:2360, DOI: 10.3390/cells10092360,
Peluso, Michael, et al.(2020). Liver function test abnormalities in a longitudinal cohort of Thai individuals treated since acute HIV infection. PLOS Pathogens, 9 pgs. PMID: 31953919.
Dagenais-Lussier, Xavier, et al.(2019). USP18 is a significant driver of memory CD4 T-cell reduced viability caused by type I IFN signaling during primary HIV-1 infection. PLOS Pathogens, 32 pgs. PMID: 31658294.
Colavita, Francesca, et al.(2018). Overproduction of IL-6 and Type-I IFN in a Lethal Case of Chikungunya Virus Infection in an Elderly Man During the 2017 Italian Outbreak. Open Forum Infectious Diseases, 21 pgs. PMID: 30539034.
Williams, Dionna, et al.(2018). CCR2 Signaling Selectively Regulates IFN-alpha: Role of Beta-Arrestin 2 in IFNAR1 Internalization. Journal of Immunology, 19 pgs. PMID: 30504423.
Zhang, Guoliang, et al.(2018). A Proline Deletion in IFNAR1 Impairs IFN-Signaling and Underlies Increased Resistance to Tuberculosis in Humans. Nature Communications, 9 pgs. PMID: 29311663.
Murayama, Goh, et al. (2017). Enhanced IFN-alpha Production is Associated with Increased TLR7 Retention in the Lysosomes of Palasmacytoid Dendritic Cells in Systemic Lupus Erythematosus. Arthritis Research & Therapy, 11 pgs. PMID: 29052537.
Wither, Joan, et al. (2017). Presence of an Interferon Signature in Individuals Who are Anti-Nuclear Antibody Positive Lacking a Systemic Autoimmune Rheumatic Disease Diagnosis. Arthritis Research & Therapy, 11 pgs. PMID: 28245862.
Vanheule, Vincent, et al. (2016). Basic Chemokine-Derived Glycosaminoglycan Binding Peptides Exert Antiviral Properties Against Dengue Virus Serotype 2, Herpes Simplex Virus-1, and Respiratory Syncytial Virus. Biochemical Pharmacology, 13 pgs. PMID: 26551597.
Combes, Alexis, et al. (2020). Global Absence and Targeting of Protective Immune States in Severe COVID-19. Nature, 36 pgs.
看到這兒,您心動了嗎?馬上聯系小艾吧!
美國的PBL Assay Science(又名:Pestka Biomedical Laboratories, Inc.)成立于1990年,創(chuàng)始人Sidney Pestka被稱為“干擾素之父”,PBL Assay Science 作為干擾素和細胞因子蛋白和抗體以及預包裝的一流的干擾素和細胞因子免疫測定試劑盒的高質量制造商而享有盛譽。生產和銷售的高品質干擾素產品和生物標志物檢測試劑盒在很多高影響力出版物中都有引用,并已在具有挑戰(zhàn)性的樣本中進行了外部驗證。
PBL Assay Science提供各種干擾素亞型的蛋白,抗體和檢測試劑盒,如:IFN-Alpha(IFN-ɑ 2a,IFN-ɑ 2b,IFN-ɑ 5,IFN-ɑ 6,IFN-ɑ 7,IFN-ɑ 14,IFN-ɑ 16,IFN-ɑ 17......),IFN-Beta(IFN-β 1a,IFN-β 1b),IFN-Lambda(IFN-λ),IFN-Omega(IFN-ω),IFN-Gamma(IFN-γ)......
武漢艾美捷科技有限公司,簡稱艾美捷,擁有進出口資質和自己的國際物流,與國內外90余家知名的試劑、原料及技術服務供應商深度合作,是國內生物制藥企業(yè)、診斷企業(yè)、跨國藥企、各大高校院所采購平臺的首選定供應商。

微信掃碼在線客服